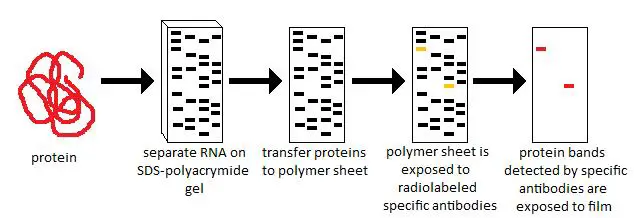
The method involves using gel electrophoresis to. To achieve this, western blot implements three steps: (1) separation by size, (2) transfer to a solid support, and (3) visualizing target protein. Western blotting is a laboratory technique used to detect a specific protein in a blood or tissue sample. Moving forward, Western blots continue to be of value in confirming results from antibody arrays, making them suitable for use in proteomics research. Western blotting is commonly used to investigate the change in abundance of a specific protein under different conditions. Western blot allows us to determine the relative protein levels between samples and establish the molecular weight of the target, which can provide insight into its post-translational processing. For example, with HIV and prion disease, Westerns are used as a key supplemental screen since their results are less ambiguous, and quicker, than other methods. In these instances, Western blots provide confirmation of results both in research and diagnostic testing. In many cases, Western blots are used in combination with other key antibody based detection techniques, such as ELISAs or immunohistochemistry. Some researchers are making existing internal loading controls more powerful by adding bioinf ormatics to the mix. Likewise, they are helpful for comparing expression of a target protein from various tissues, or seeing how a particular protein responds to disease or drug treatment. Several recent studies hint at the future of Western blot normalization and analysis. Their ability to clearly show the presence of a specific protein both by size and through the binding of an antibody makes them well-suited for evaluating levels of protein expression in cells, and for monitoring fractions during protein purification. Western blots are in wide use across a broad range of scientific and clinical disciplines. Lanes 2 & 3: Common alterations that modify the molecular weight Lane 1: Target protein band at the expected molecular weight. Alternate splicing may also cause unexpected size variations, as may the particular combination of charged amino acids found in the protein. Is the band of lower or higher molecular weight than expected? Is there a single band, or are there several bands? A smaller than expected band could indicate that the protein has been cleaved or is degraded.Ĭonversely, when bands are seen at higher levels than expected, this may indicate an actual increase in mass due to glycosylation or multimer formation. Since proteins are separated by size during the gel electrophoresis stage, and then detected by a specifically directed antibody, the procedure essentially confirms the identity of a target protein.įurthermore, when data does not match expectations, there may be clues as to what should be investigated to determine the reason. Western blotting is an extremely powerful technique, despite its overall simplicity, because it provides additional information not readily gathered from other key immunological laboratory techniques.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
